Research
How do we make functional tissues?
Tissues are made up of diverse, specialized cell types that work together to maintain homeostasis and function. To do this, cells must adapt to constant environmental changes—like nutrient levels, stress, and time. This challenge becomes even greater during embryonic development, when tissues are being generated. Here, populations of stem cells must interpret their signalling environment, adopt specific identities, and move into place to ensure the proper development of functional tissues. Further, these physical changes in tissue structure (morphogenesis) occur alongside internal gene regulatory shifts that define cell fate, posing the question: How do physical morphogenesis and gene regulation interact?
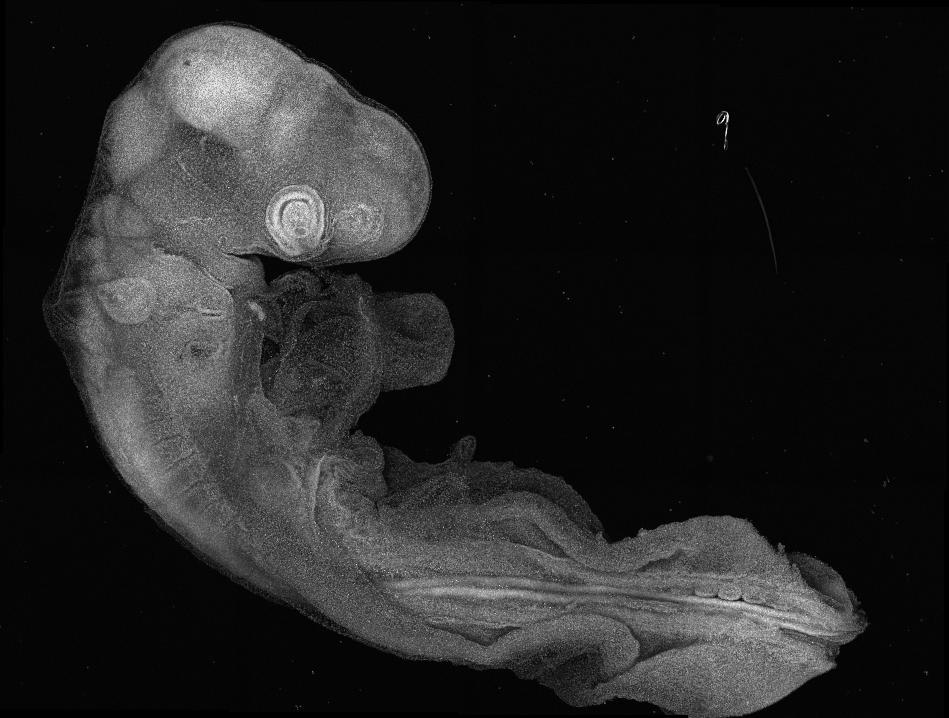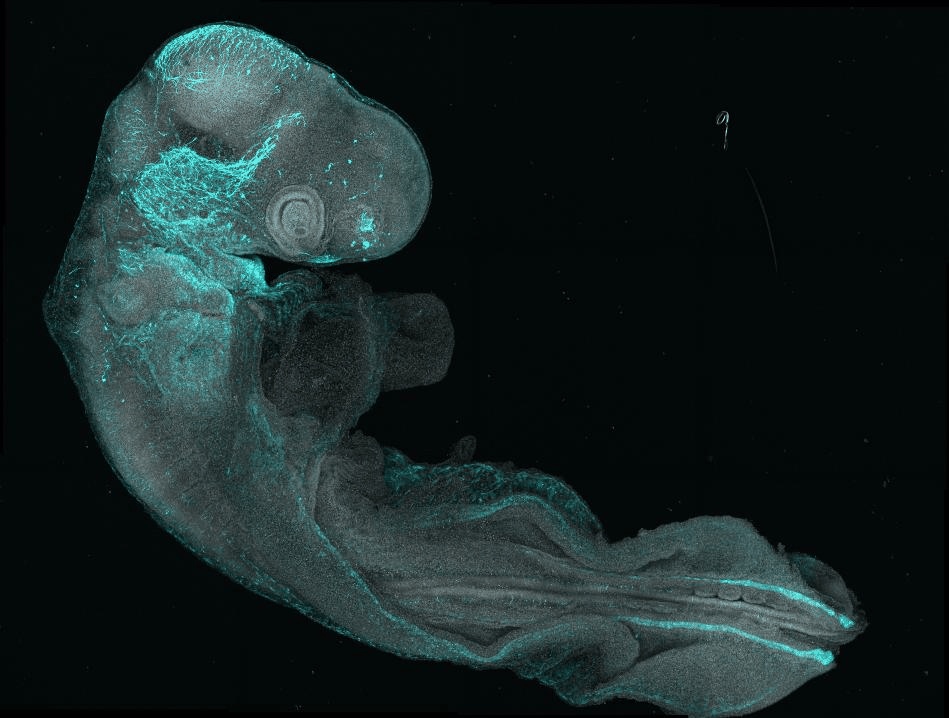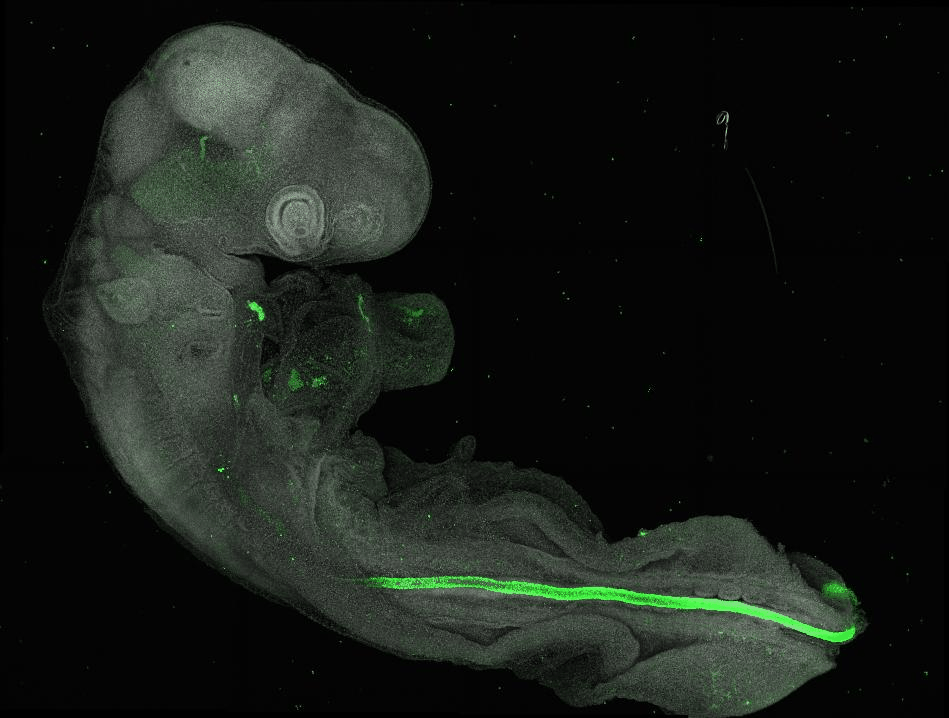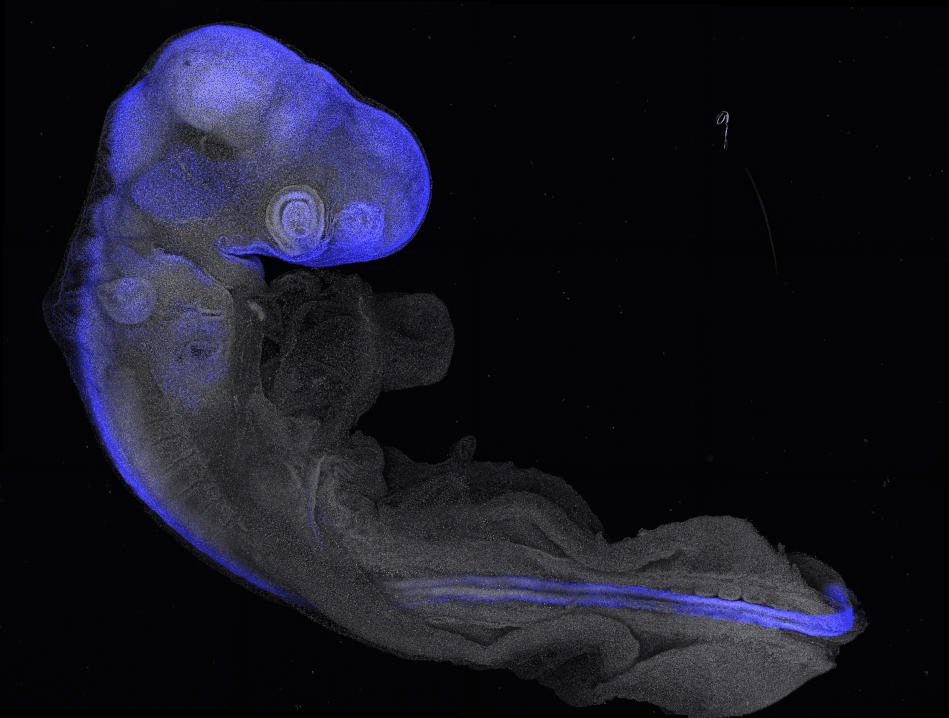
Our lab addresses this by studying how stem cells maintain a permissive epigenetic state to precisely control gene expression and fate decisions amid changing signals and tissue shapes. Using the developing neural tube as a model—an organ that undergoes extensive morphogenesis and generates diverse nervous system cell types—we explore the feedback between intrinsic cell states and their environment.
By combining in vivo studies in chick embryos with in vitro human stem cell models, we investigate tissue formation across scales to reveal key developmental mechanisms and inform tissue engineering and regenerative medicine.
Ongoing projects
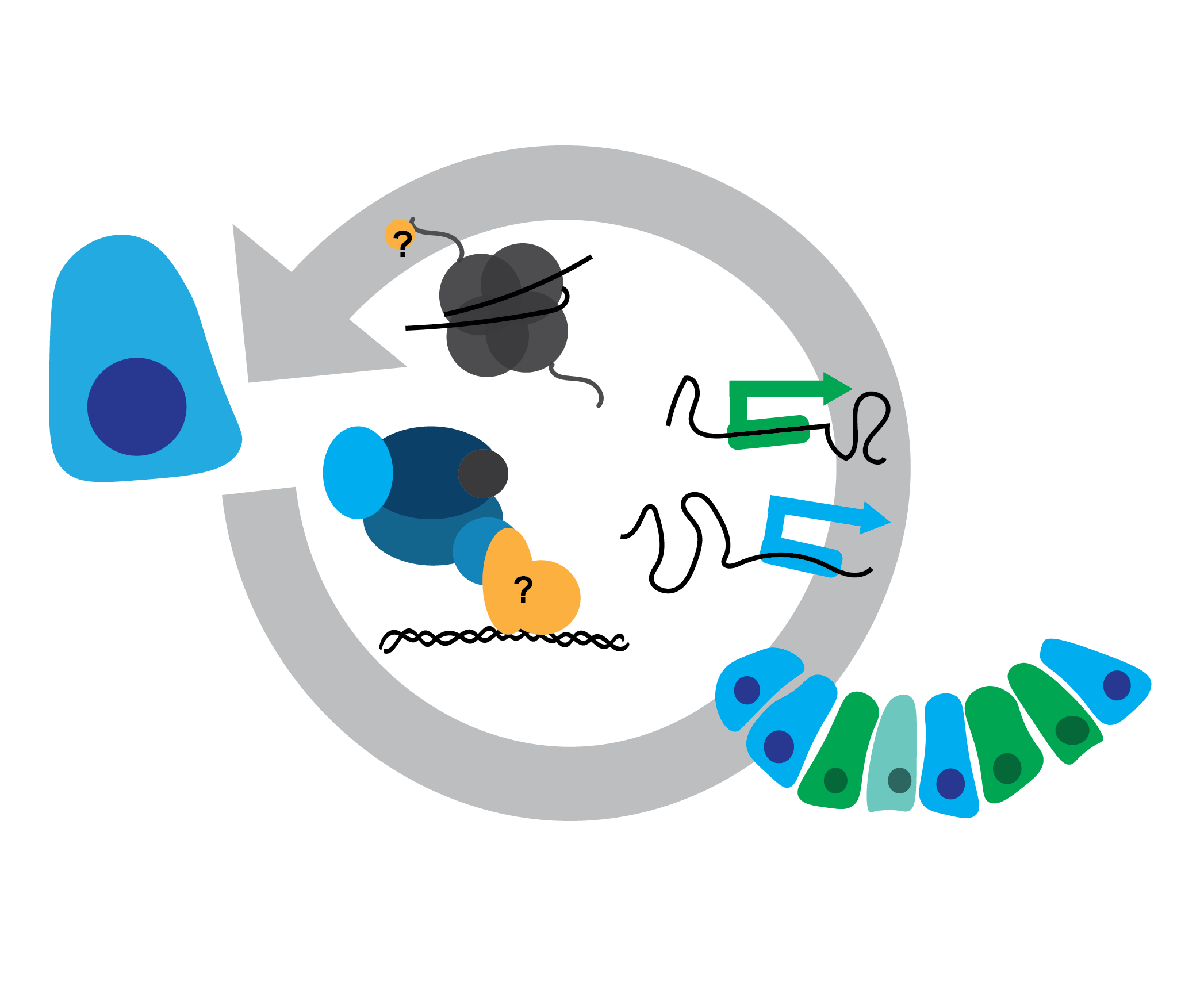
Connecting transcription to environment
One way cells interpret complex external environments is by maintaining an epigenetic state poised to respond to signals across cellular and tissue levels. Neural tube progenitors use bivalently marked genes to rapidly initiate transcription in response to signals and inform cell fate. But how does this occur? What epigenetic machinery needs to be in place? How many morphogenic signals can be interpreted at once?
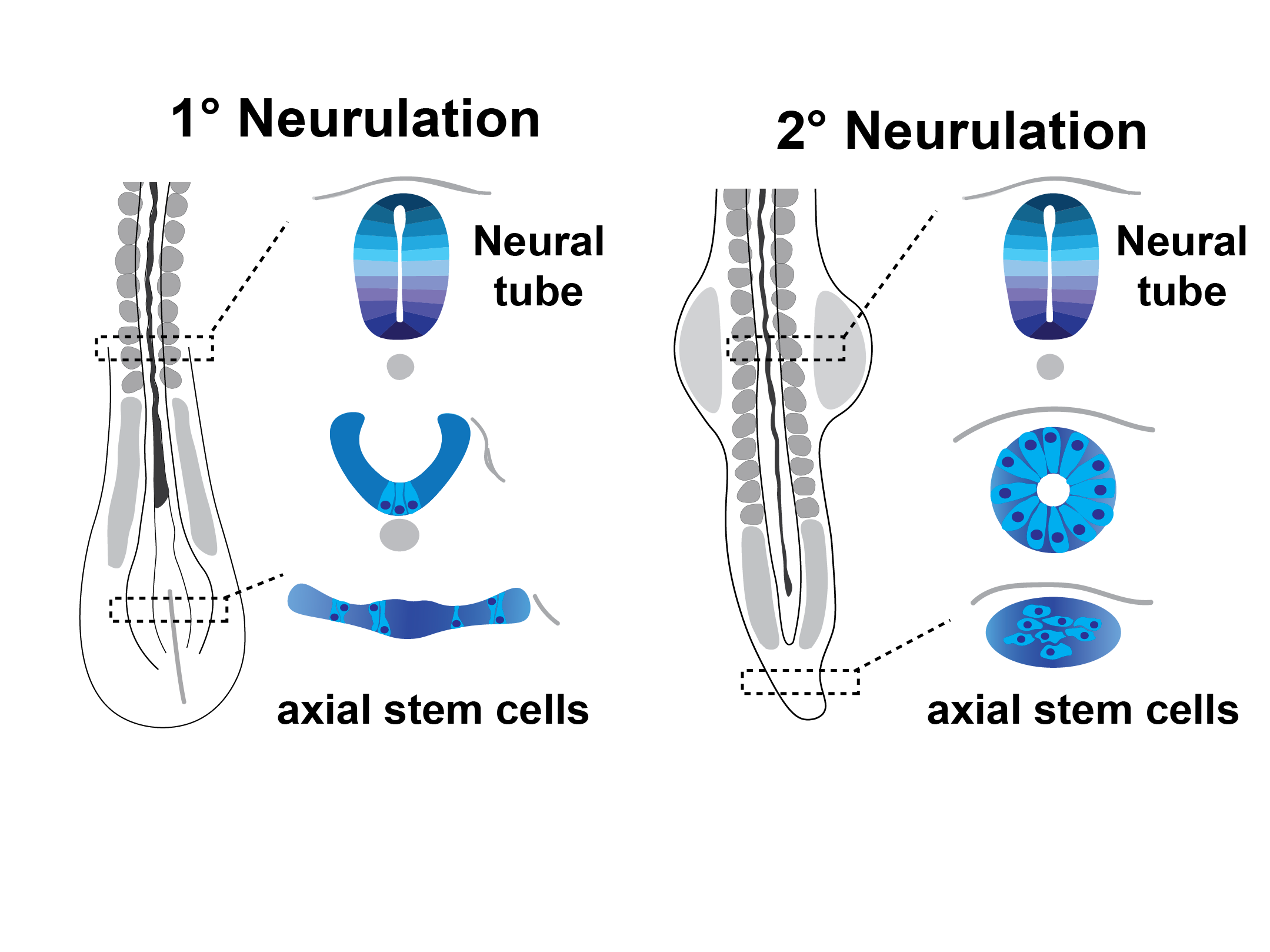
Buffering against changing environments
The neural tube forms through two morphogenetic processes depending on developmental timing: early primary neurulation and later secondary neurulation. In the former, stem cells roll from an epithelial plate into a tube; in the latter, they condense into a cord, epithelialize, and form a lumen. Both cases result in the acquisition of neural fate and formation of a tube. This raises key questions: How do cells ensure the robust formation of a tube from different starting points? How are the same cell types made and organized properly? Can we use this knowledge to build tissues in a dish?
